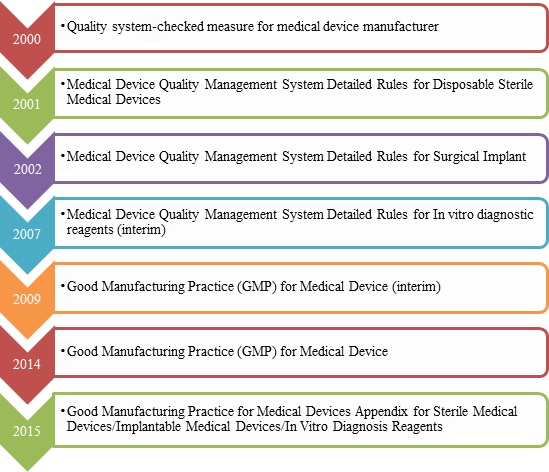
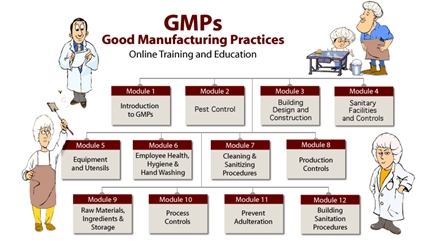
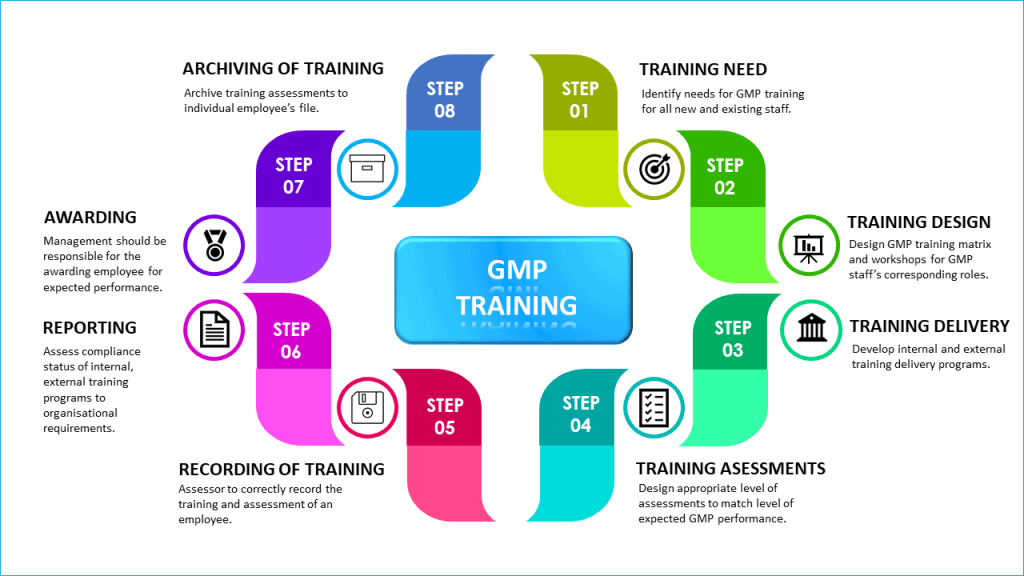
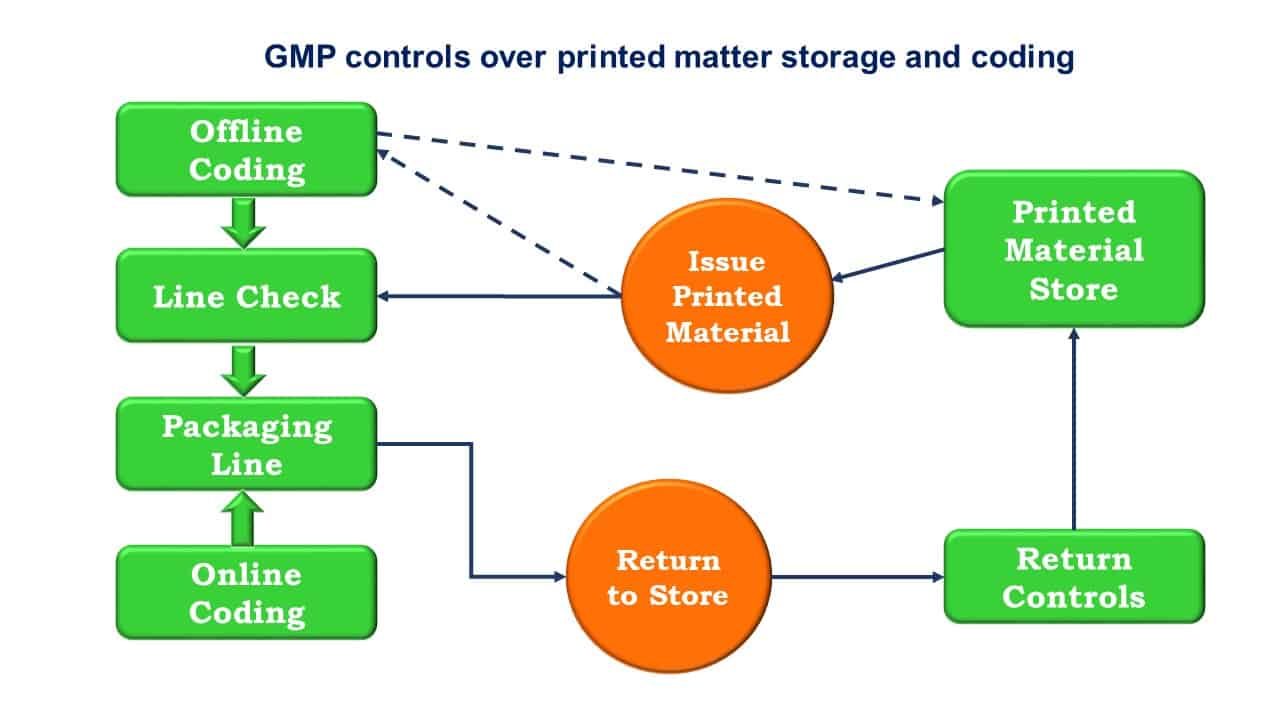
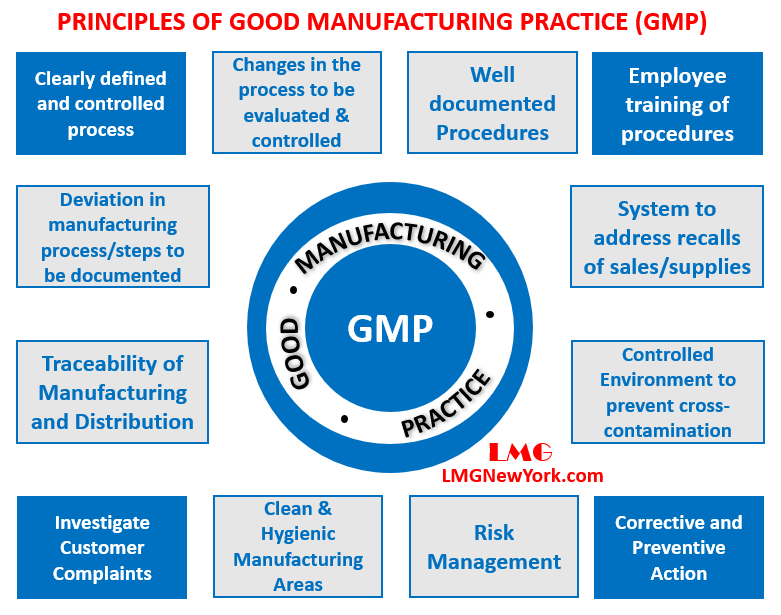
Quality Transintegra on Twitter: "#WHO #GMP Training Basic GMP principles for production areas https://t.co/LTp6C8K83c" / Twitter

Pharmaceutical Quality Control Lab : GMP (Good Manufacturing Practices) Training for Pharmaceutical Manufacturing, Covering FDA Regulations of Laboratory Results, Sop's (Standard Operating Procedures) and Oos (Out of Standard) and Oot (Out of